AlertsNet2.0
Incidence of bloodstream infections, antibiotic resistances, and blood culture ordering and testing practices – a Thuringia-wide prospective population-based surveillance and registry

The current failure of new approaches to the treatment of blood stream infections (BSI) is closely linked to the deficits of differentiated diagnostic options. The timing of diagnosis and thus the early initiation of therapeutic measures is, however, the decisive determinant of high lethality. The detection of microorganisms by the blood culture (BC) method is an indispensable prerequisite for an adequate antimicrobial therapy of sepsis. The knowledge of the pathogen and its sensitivity to antibiotics allows targeted anti-infective measures after initiation of an empirical initial therapy and sets the course for further diagnostic procedures. This improves the prognosis, shortens period of hospitalization and helps to avoid an anti-infective over-therapy.
The implementation of blood culture diagnostics according to current guidelines is therefore an important prerequisite for a successful therapy of septic patients. In only 10% of patients with severe sepsis, pathogen detection is successful. Long transport times of the BC bottles from the intensive care unit to the microbiological laboratory contribute decisively to this situation. Furthermore, the number of blood cultures is too low (16.3 BC sets / 1,000 patient days in Germany1). According to an analysis of data from the hospital infection surveillance system (KISS) from 223 German intensive care units, at least 90 BC sets / per 1,000 patient days should be taken from intensive care units2. For Germany, population-related data on antibiotic resistance are scarce and clinical patient-related data on the demographic factors morbidity and lethality are not available. On the other hand, population-based data on BSI rates with patient references from surveys of the International Bacteremia Surveillance Collaborative (IBSC)3 are available from Canada, Scandinavia and Australia.
The Thuringia-based prospective population-based AlertsNet register establishs a data collection and alerting system for the diagnosis of BSI, identifying risk factors, and improving the treatment and outcomes of hospitalized patients with BSI. The project uses a nationally established electronic database for BC results. In addition, the database contains clinical and demographic data from patients with a confirmed BSI. From ~ 316,000 BC sets expected per year, ~ 25,300 BC sets will be positive, of which approximately ~ 20,200 will be clinically relevant positive (without contamination), corresponding to about 5,000 patients. Primary endpoints are the incidence rate of positive blood cultures (number / 1,000 BC sets) and the total number of collected BC sets (number / 1,000 patient days and sending institution).
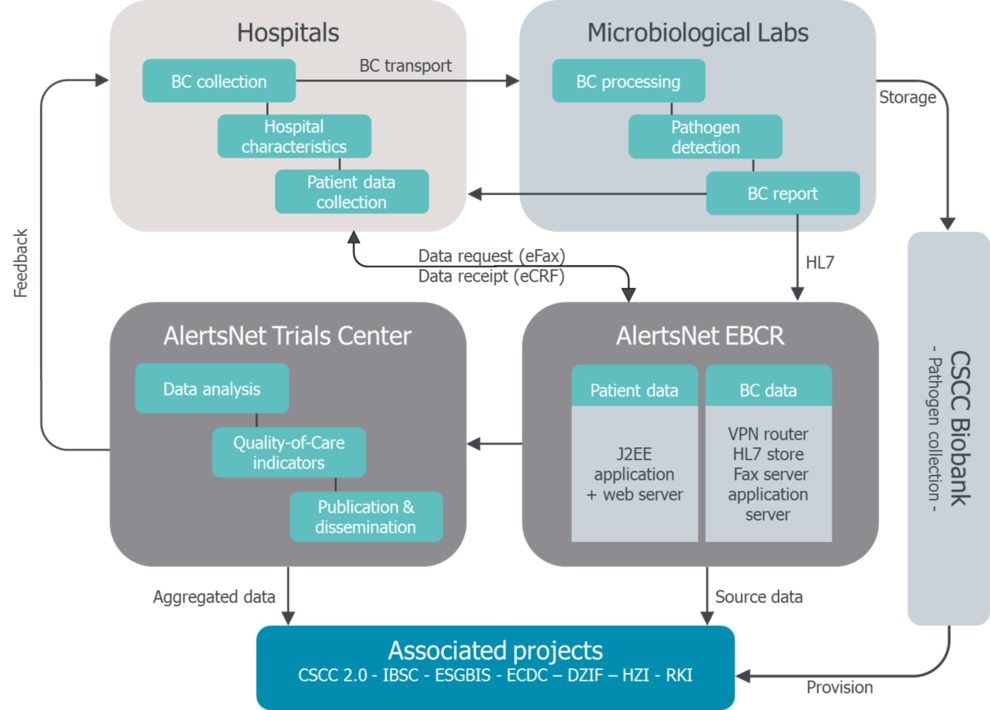
The collected data allow to develop quality indicators and to determine reference values for these indicators after adjustment of the hospital characteristics as well as an analysis of the relationship between the incidence rate of BC taking, case mix, disease severity, lethality and the size of the hospital.
By 2018, the network will combine up to 38 hospitals and 18 microbiological laboratories, thus providing population data for the State of Thuringia. A transfer of the data to EARS and an exchange with the IBSC is provided.
- EARS-Report 2013/2014, European Antimicrobial Resistance Surveillance System, www.ecdc.europa.eu
- Karch A, Castell S, Schwab F, Geffers C, Bongartz H, Brunkhorst FM, Gastmeier P, Mikolajczyk RT: Proposing an empirically justified reference threshold for blood culture sampling rates in intensive care units. J Clin Microbiol 2015; 53(2): 648–52
- Laupland KB, Schønheyder HC, Kennedy KJ, Lyytikäinen O, Valiquette L, Galbraith J, Collignon P, Church DL, Gregson DB, Kibsey P: Rationale for and protocol of a multi-national population-based bacteremia surveillance collaborative. BMC research notes 2009; 2: 146


