Principal investigator: Prof. Dr. Carsten Hoffmann
DFG TRR 166 2015-2020
Projektleiterin: Prof. Dr. Britta Qualmann
DFG 2020-2020
Ligand receptor interaction is often simply seen as a binding event and neglects the conformational dynamic of the target protein and its influence on ligand binding or the efficacy of the ligand. Within this project will measure conformational dynamic of a receptor and monitor ligand binding with high kinetic resolution in living cells. We will use a fluorescence-resonance-energy-transfer (FRET) based approach to measure ligand on/off-rates and receptor activation/deactivation in real-time to obtain ligand residence time information and ligand efficacy data in living cells.
Principal investigator: Prof. Dr. Regine Heller
DFG RTG 2155 2016 - 2020
Posttranslational protein modifications (PTMs) of proteins represent molecular mechanisms, which may lead to age-dependent functional decline of cells and tissues. The current project investigates modifications of AMPK, an important metabolic sensor and regulator enzyme, in vascular endothelial cells. We propose that PTMs of AMPK and AMPK-regulating enzymes lead to endothelial dysfunction thereby supporting the development of age-associated vascular diseases. The aim of this work is to identify these PTMS and to characterize AMPK as a pharmacological target to protect from vascular ageing.
Further information
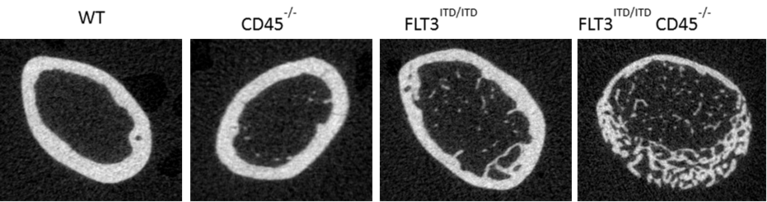
Principal investigator: Prof. Dr. Reinhard Bauer
DFG RTG 1715 2018 - 2021
Mechanisms mediating the balance of resistance and tolerance responses of the host to infectious stress are largely unknown. Preliminary data disclose PI3Kg as a mediator of memory-like adaptive responses in microglia. The current approach intends to clarify PI3Kg -related signaling processes involved in microglial adaptive responses controlling trained sensitization and tolerance. Aim of this approach is to decipher whether or not these microglial adaptive responses are able to ensure a specific impact on inflammatory preconditioning in focal brain ischemia.
Further information
Principal investigator: Prof. Dr. Reinhard Bauer
BMBF 2018 - 2021
Schwann-cell tumors (SCT) are benign tumors of peripheral nerves. SCT grow non-invasively but displacing and disturb therefore the affected nerves. Until now, no specific therapy is available. Aim of this project is to perform a preclinical confirmatory study (PCS). This PCS is mandatory in order to verify that the intended translation of the novel rhNRGβ1 protein substitution therapy for SCT treatment in humans is accomplished on a secured scientific basis.
Subproject
Conformational signature of CXCR4/ACKR3 activation in β-arrestin-1 and -2
Principal investigator: Prof. Dr. Carsten Hoffmann
EU 2020 - 2014
Our current knowledge is very limitted with respect to GPCR/arrestin interaction and the role of individual GRKs in this process. Most of the GPCR-family members are regulated by only four of the seven members of the GRK-family (namely GRK 2,-3,-5, and -6) and two of the four arrestin proteins that exist in human physiology, namely β-arrestin 1 and β-arrestin 2. For this system of GPCR regulation to be conceivable, β-arrestins require the ability to adopt to a multitude of different receptor activation and phosphorylation states, supposedly facilitated by a high degree of conformational plasticity within β-arrestin.
Further information