A disruption of epithelial and endothelial barrier function is a typical pathological change in acute sepsis. Two major mechanisms have to be considered responsible for barrier breakdown. Signals generated by the deregulated and overreacting immune system, as well as signals or activities from pathogenic bacteria and fungi directly interacting with epithelial or endothelial cells are assumed to contribute to barrier breakdown. Here we want to specifically analyze inactivation of the apical junctional complex (AJC) and the role of junction-associated adaptor proteins with NACos (nuclear and adhesion complex) function (e.g. catenins, ZO-proteins) during sepsis. Both pathways are interconnected and provide interesting targets to treat organ failure in sepsis. Microfluidically supported biochips will be used to study the cross-communication of human gut and liver tissue models with respect to the impact of cytokine-mediated regulation of tight and adherens junction protein function in the breakdown of endothelial and epithelial barrier and subsequent hepatocellular dysfunction.
Sepsis is the predominant cause of multi-organ failure, which determines prognosis in the critically ill. The current concept assumes that end organ failure results from an uncontrolled inflammatory response; yet, strategies to neutralize mediators of inflammation have failed to improve outcome in numerous trials. Mounting evidence suggests that cell type-specific activation and control of signaling events mediate development or resolution of organ dysfunction. Projects in the research area Damage address sepsis-induced remote organ failure with a special focus on the liver. The ultimate goal is to identify and explore potential novel “drugable” targets, such as signaling or barrier failure, to cell- and tissue-specifically interfere with the underlying pathophysiology. We aim to efficiently translate promising results obtained during the first funding period to preclinical testing. Systematic cooperation with the Research Campus InfectoGnostics, the Jena Center for Soft Matter, and the Center for Innovation Competence Septomics to develop companion diagnostics and theragnostic strategies will help us to avoid the common discrepancy between success in animal testing and failure of adjunctive interventions in clinical trials.
The core of this research area is defined by six translational projects:
Targeted strategies in organ failure | Damage
G
GuliverMacrophage Polarization and Sepsis-related Inflammasome Regulation in Gut and Liver Organoids

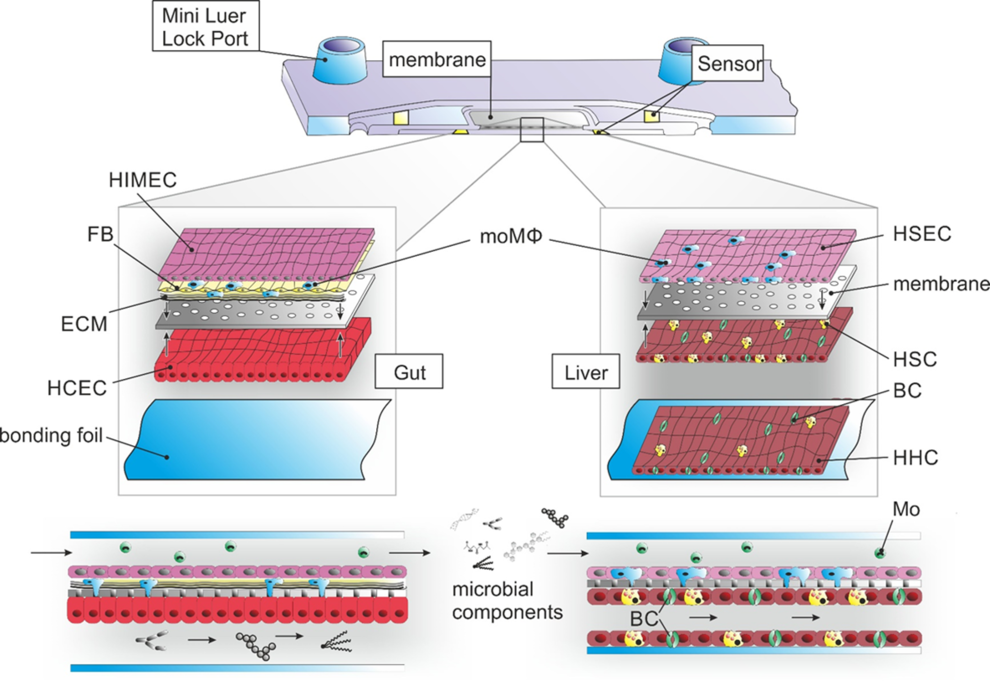
I
InsultLiverConsequences of NLRP3 inflammasome activation in septic liver injury
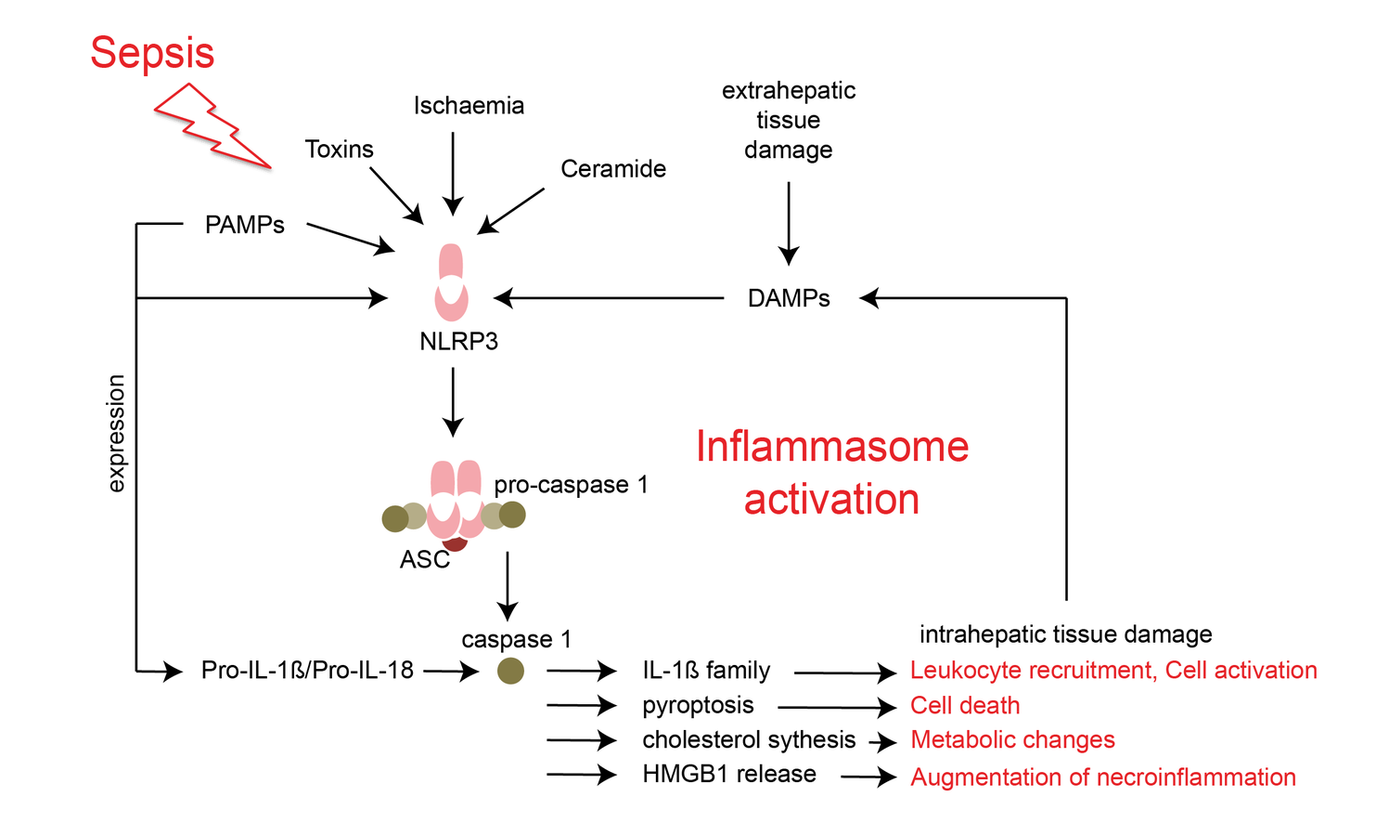
The inflammatory immune response during sepsis is a double-edged sword, which supports pathogen elimination on one hand but may induce and aggravate organ failure on the other hand. The activation of inflammasome complexes is crucial for responding to invading pathogens during sepsis but mounting evidence suggests that unrestricted inflammasome activation plays a central role in perpetuating organ damage in a variety of diseases. Hepatic activation of NLRP3 induces inflammation, fibrosis, and cell, but its regulation in septic liver injury is poorly understood.
In this project we will investigate the regulation of the NOD-like receptor NLRP3 during septic liver injury and its consequences on hepatic inflammation, metabolism, and cell death. We will study inflammasome activation in different cell types using the model of polymicrobial abdominal sepsis. The use of molecular multimodal imaging will allow to investigate the consequences of organ injury in a fast and label-free fashion over large areas of the liver in real-time. In addition, we will investigate, whether - and what kind of - soluble factors mediate inflammasome activation in patients with septic liver injury.
The overall aim of the project is to understand time- and cell-specific regulation of inflammasome activation during the course of sepsis in order to provide the opportunity of delivering targeted anti-inflammatory strategies to improve the outcome of patients with septic liver injury.
N
NanoDyeLiveryHepatocyte-specific targeting using nanoparticles

The liver plays a pivotal role in the host response to life-threatening infections. Its capacity to excrete toxic substances via the bile appears exceptionally sensitive to inflammation, a hallmark of life-threatening infections. From a molecular point of view, the failure to produce bile can be explained by down-regulation of transport proteins at the plasma membrane of liver cells facing the bile canaliculi. In our previous work we have shown that failure to produce bile and excrete toxic compounds from the liver is a common problem, portends poor outcome and depends on pluripotent signaling molecules. Thus, mice lacking these signaling molecules are protected against such negative effects.
However, these molecules also play a crucial role in the recruitment of immune cells to the focus of infection and in their ability to kill bacteria. As unselective suppression of such effector molecules and the concomitant severe immune defect outweigh the beneficial effect on liver function. NanoDyeLivery attempts to unravel the mechanisms of signal molecule-dependent dysfunctions of the bile-producing machinery and to specifically modulate such molecules in the liver while preventing off-target effects on the immune system.
Nanoparticles have been proven to offer unique properties to overcome cellular barriers in order to improve delivery of drugs. We have observed that the fluorescent dye DY635 selectively enters liver cells and used this finding to design hepatocyte-specific nanoparticles. As immune cells do not take up the drug-loaded particles, we will address whether the modulation of intracellular signaling molecules prevents the failure to produce bile without a concomitant impairment of the immune system and thereby enable a novel treatment option.
Macromolecular innate immune complexes in neonatal sepsis
In neonates the response of the innate immune system is critical for the initiation and maintenance of host defense. Altered inflammatory conditions, e.g. meningitis, are closely associated with neonatal sepsis. Gram negative bacteria such as E. coli and Gram positive bacteria such as Group B streptococci (GBS) induce the formation of cytokines by bacterial LPS and RNA respectively. GBS RNA instructs cytoplasmic macromolecular complex formation of Myeloid differentiation primary response gene 88, Toll-like receptors and inflammasomes containing inflammatory caspase.
Our project focuses on evidences of altered innate immune response during septic organ damage. We will experimentally analyze control mechanisms and regulations of multiprotein innate immune complex formation leading to the characteristic organ damage related inflammatory condition. We want to study properties of the innate immune complex as biomarker for neonatal sepsis and as an anti-inflammatory drug target. Furthermore, molecular determinants of complex formation and translocation across biological barriers will be studied. Results hence obtained will be translated for preterm born and very low birth weight infants at high risk for sepsis.
T
TarOrgSterolTargeting the cholesterol-pathway to prevent pulmonary and remote organ dysfunction in pneumococcal disease

Lehrstuhl für Organische und Makromolekulare Chemie (IOMC), Center for Soft Matter (JCSM), Friedrich-Schiller-Universität Jena
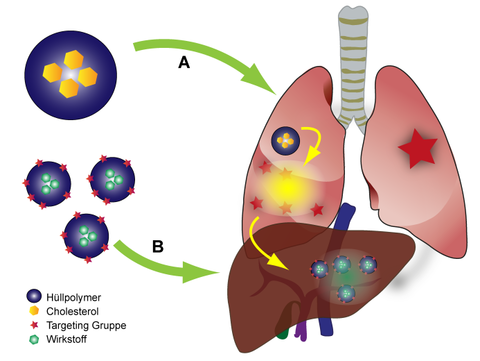
Pneumonia reflects a frequent clinical condition that may lead to lung barrier failure, bacteremia, and sepsis, including local (ARDS) and remote organ dysfunction. Underlying infections are often caused by Streptococcus pneumoniae, a Gram-positive pathogen that produces pneumolysin as a virulence factor. Pneumolysin, a cholesterol-binding pore-forming toxin, may trigger barrier failure and the onset of sepsis. We have shown that changes in sterol biosynthesis in cells of the liver (hepatocytes), in particular cholesterol biosynthesis, are induced as a remote adaptive response to circulating pneumolysin. Indeed, a communication between the infection of the lung and the liver occure. It was shown that an increased level of cholesterol in human is associated with a reduced severity of infections. The therapeutic potential of interfering pathways is unexplored. However, even simple delivery of cholesterol, a detergent, to the lungs poses significant hurdles. We propose to design and monitor interventions to prevent local and remote organ failure using cutting-edge technologies, such as recently developed organ-specific nanoparticles, tranporting drug to hepatocytes, and systems biology approaches for transcriptome and metabolome analysis, respectively, to answer whether selectively targeting the cholesterol pathway can improve lung function during pneumococcal disease and prevent septic disease progression and remote organ failure.
Targeting endothelial barriers in sepsis


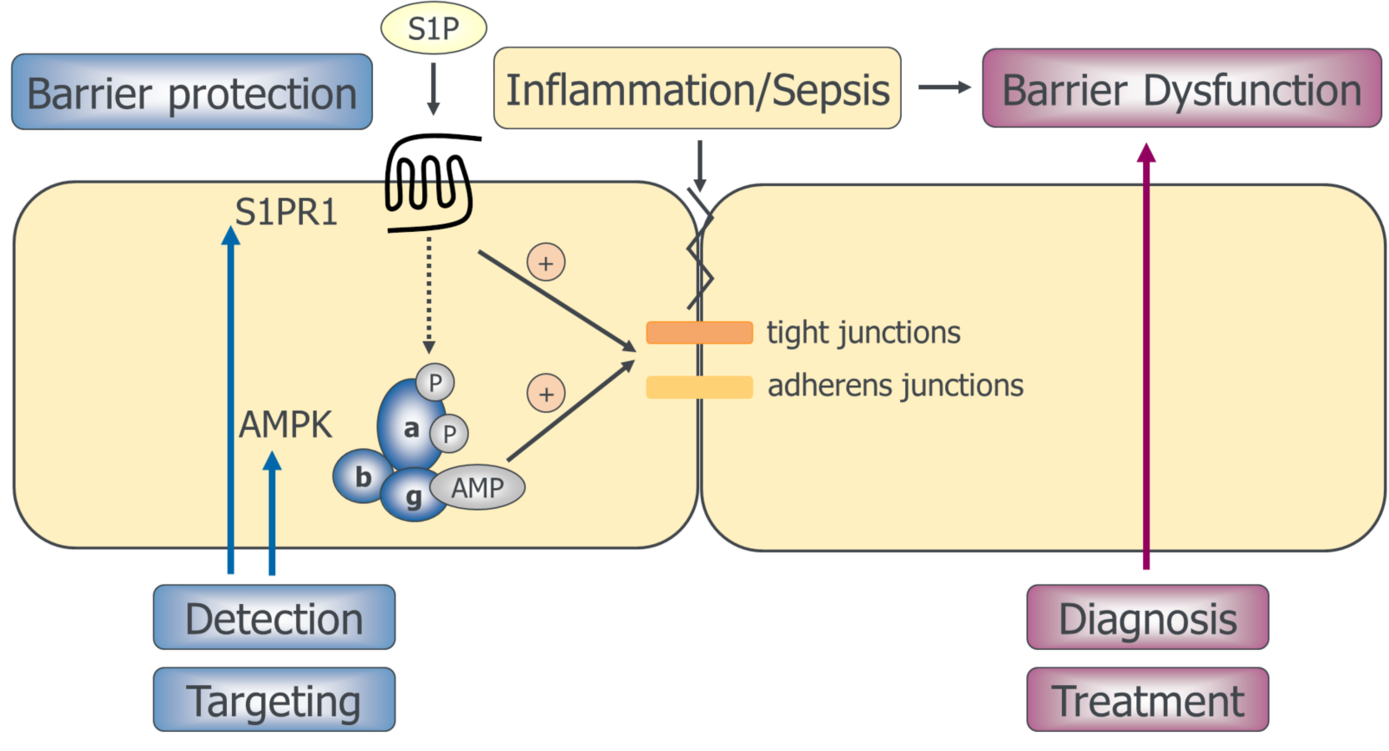
Microvascular dysfunction, a critical hallmark of sepsis that results in decreased organ perfusion and subsequent development of organ failure, is characterized by disruption of endothelial barriers. An early diagnosis of barrier dysfunction, followed by preventive or therapeutic strategies, may therefore improve sepsis outcome. The current project investigates two interrelated barrier-protecting pathways, the sphingosine 1-phosphate (S1P)/S1P receptor type 1 (S1PR1) axis and the AMP-activated protein kinase (AMPK) pathway, to elucidate their possible diagnostic and therapeutic value in the context of sepsis. We will establish diagnostic tests to determine the status of S1P/S1PR1 signaling and AMPK activity in septic patients in a prospective observational study. In this context, a unique proprietary monoclonal antibody that detects human S1PR1 on the cell surface by flow cytometry (FACS) will be applied. Correlation of AMPK phosphorylation and S1P/S1PR1 signaling with established biomarkers of vascular dysfunction and organ dysfunction will reveal their role in the complex pathophysiology of sepsis. Furthermore, targeting strategies for both pathways will be investigated in experimental models. The proposed project will be the first study exploring the clinical relevance and potential therapeutic use of S1P/S1PR1 signaling and AMPK activation for endothelial barrier maintenance and stabilization in septic patients.






